Phytochemical Profiling and Antioxidant Assessment of a Herbal Formulation Containing Carica papaya, Azadirachta indica, and Allium sativum
| Received 29 Dec, 2025 |
Accepted 12 Mar, 2026 |
Published 20 Mar, 2026 |
Background and Objective: Whole plants or combinations of plants and herbal extracts are frequently used as therapeutic preparations in traditional systems that take a holistic approach to health and illness. The biologically active secondary metabolites of plants found in roots, leaves, flowers, stems, and bark are called phytochemicals, and they have several pharmaceutical uses in human systems. The present study evaluated the phytochemicals and antioxidant potentials of the herbal formulation of Carica papaya, Azadirachta indica and Allium sativa. Materials and Methods: The herbal formulation was prepared by combining the pulverized samples of Carica papaya, Azadirachta indica, and Allium sativa in the ratios (1:1:1, 2:1:1, 1:2:1, and 1:1:2 w/w), respectively, to obtain four permutations, which were extracted using ethanol. Phytochemicals and antioxidant potentials evaluations were carried out utilizing the extracts obtained. Data were analyzed using ANOVA in SPSS version 20 (SPSS Inc., Chicago, IL, USA), with Duncan’s multiple range test for post-hoc comparisons; results are expressed as Mean±SD, significance was set at p<0.05, and charts were prepared using Microsoft Excel. Results: Total phenolic content shows a decreasing trend as D>C>B>A (149.5>98.25>20.78>8.47 GAE mg/100 g) across the different ratios. Also, a similar trend was noticed with the flavonoid content, D>C>B>A (271.95>178.77>37.91>15.54 QCE mg/100 g) across the different ratios. The presence of tannins and saponins did not vary greatly across the different ratios of the formulation. All the ratios showed reasonable ferric reducing antioxidant power (FRAP), hydrogen peroxide and anti-lipid peroxidation inhibitions capacities. Conclusion: The various ratios of ethanol extract of herbal formulation containing Carica papaya, Azadirachta indica and Allium sativa exhibited pronounced antioxidant activity supported by the presence of potent free radical scavenging and biologically active compounds.
| Copyright © 2026 Abu et al. This is an open-access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. |
INTRODUCTION
Due to their possible pharmacological effects, medicinal plants are now the subject of extensive research. In fact, several clinically beneficial therapeutic agents that are essential in the treatment of human illnesses have been found as a result of the search for novel pharmacologically active molecules through the screening of natural sources and plant extracts. Nowadays, 80% of people in industrialized nations utilize traditional medicine, which contains substances made from therapeutic herbs1. Due to the wide range of chemical ingredients available, natural products, whether as pure chemicals or as standardized plant extracts, provide countless options for novel therapeutic lead molecules. Papaya (Carica papaya), neem (Azadiracta indica) and garlic (Allium sativum) have long been used in traditional medicine and food as anti-infective agents2.
The leaves of the Carica papaya have a long history of medical usage and have been used to cure a variety of illnesses in many Asian and African nations. Corns, warts, constipation, weakness, amenorrhea, menstrual issues, eczema, sinusitis, cutaneous tubercle, glandular tumor, diabetes, ulcers, hypertension, dengue, etc. are all treated with them due to the existence of the previously indicated significant functional elements3. Indigenous Australians have long used papaya leaf extract for its anti-cancer properties4. The leaves of C. papaya are rich in nutrients that boost immunity in addition to their many cancer-fighting properties. In addition to vitamins E, A, and C, they contain vitamin B-17, whose concentrated version is utilized in traditional chemotherapy treatments to cure cancer patients. Papaya leaf phytochemicals have been shown to stop platelet breakdown and bone marrow depletion5. Papaya leaf juice can help normalize blood coagulation and mend the liver by increasing the platelet count and red and white blood cells6.
In medical folklore, the neem tree including its leaves, bark, fruit, flowers, oil, and gum is used to treat a number of illnesses, including diabetes, cancer, high blood pressure, and heart disease7. Cellular and molecular mechanisms may be responsible for the potential effects observed when using these extracts. These mechanisms may include immune surveillance, detoxification, DNA repair, cell cycle alteration, free radical scavenging, anti-inflammatory, anti-angiogenic, and anti-metastatic activities, as well as the capacity to modulate different signaling pathways8,9.
One of the most sought-after botanicals, garlic is a fragrant herb and spice made from tubers that is used in both cooking and traditional medicine to treat a wide range of illnesses worldwide. Previous investigations have demonstrated a variety of pharmacological characteristics of this species, including its anticancer, antibacterial, anti-inflammatory, anti-hyperlipidemic, antioxidant, hypoglycemic, and hepatoprotective properties10. Allicin, diallyl disulfide (DADS), vinyldithiins, ajoenes (E-ajoene, Z-ajoene), diallyl trisulfide (DATS), micronutrient selenium (Se), and other sulfur-containing phytochemical substances are found in Allium sativa. Modulations in its antioxidant activities are associated with organosulfur compounds11,12. The chemicals found in garlic have also been reported to serve as strong immunostimulants or as promising immune-boosters. Cardiovascular conditions, cancerous growth, rheumatism, diabetes, intestinal worms, flatulence, colic, dysentery, liver diseases, facial paralysis, TB, bronchitis, hypertension, and a number of other illnesses are all treated by A. sativa10.
In the present study, we evaluated the phytochemicals and antioxidant of herbal mixture of Carica papaya, Azadirachta indica and Allium sativum.
MATERIALS AND METHODS
Study area: The study was conducted at Federal University Wukari, Taraba State. Wukari is a town and Local Government Area (LGA) in the Southeastern part of Taraba State, Nigeria, situatedalong the Katsina-Ala road, near the Benue River Basin, known as a historic Jukun kingdom center. It is approximate coordinates are 7°51'N Latitude and 9°47' E Longitude. This study was conducted over a period of 10 months, from November, 2024 to August, 2025.
Collection and identification of plant materials: Matured and healthy-looking leaves of Carica papaya (Pawpaw), Azadirachta indica (Neem) and Allium sativa (garlic) bulb were collected within Wukari, Taraba State, Nigeria. The plants were identified and authenticated at University of Agriculture Makurdi, Benue State where a voucher was deposited.
Preparation and extraction of herbal formulation: The collected plant materials were rinsed in clean water and air dried at room temperature under the shade to constant weight. The dried leaves were pulverized into powder using Thomas-Wiley laboratory mill (model 4) before being extracted by cold maceration. The herbal formulation was made by combining the pulverized samples of Carica papaya, Azadirachta Indica and Allium Sativa parts in the ratios (A-1:1:1, B-1:1:2, C-2:1:1, and D-1:2:1) respectively to obtain four permutations of the herbal formulation.
Exactly 500 g of each of the herbal formulated ratio was suspended in 2.5 L of absolute ethanol and the solution was left standing for 72 hrs in large amber bottles with intermittent shaking. At the end of the extraction, the crude ethanol extract was filtered using Whatman No. 1 filter paper (1 mm mesh size) and concentrated using water bath maintained at 45°C until dark residue was obtained. The concentrated ethanol extract was stored in an air-tight sample container in a refrigerator for further analysis.
Determination of total phenolic content: Total phenolic content was estimated by Folin Ciocalteu’s method as described by Molole et al.13.
A 1 mL of aliquots and standard gallic acid (10, 20, 40, 60, 80, 100 μg/mL) was positioned into the test tubes and 5 mL of distilled water and 0.5 mL of Folin Ciocalteu’s reagent was mixed and shaken. After 5 minutes, 1.5 mL of 20 % sodium carbonate was added and volume made up to 10 mL with distilled water. It was allowed to incubate for 2 hours at room temperature. Intense blue colour was developed. After incubation, absorbance was measured at 750 nm spectrophotometer using UVvisible Jasco V-630 instrument. The extracts were performed in triplicates. The blank was performed using reagent blank with solvent. Gallic acid was used as standard. The calibration curve was plotted using standard gallic acid. The data for total phenolic contents of C. maxima leaves were expressed as mg of gallic acid equivalent weight (GAE)/100 g of dry mass.
Determination of total flavonoid: Total flavonoid content was measured with the aluminium chloride colorimetric assay described by Kalita et al.14.
One mL of aliquots and 1 mL standard quercetin solution (100, 200, 400, 600, 800, 1000 μg/mL) was positioned into test tubes and 4 mL of distilled water and 0.3 mL of 5% sodium nitrite solution was added into each. After 5 minutes, 0.3 mL of 10% aluminium chloride was added. At 6th minute, 2 mL of 1 M sodium hydroxide was added. Finally, volume was made up to 10 mL with distilled water and mix well. An orange-yellowish colour was developed. The absorbance was measured at 510 nm using a UV-visible Jasco V-630 spectrophotometer. The blank was performed using distilled water. Quercetin was used as a standard. The samples were performed in triplicate. The calibration curve was plotted using standard quercetin. The data of total flavonoids of the leaf powder were expressed as mg of quercetin equivalents/ 100 g of dry mass14.
Determination of tannins: Sharma et al.15 made modest adjustments to Swain’s (1979) approach for the quantitative measurement of tannins. In a parafilm-covered beaker with 20 mL of 50% methanol, finely ground C. maxima leaves were heated to 80°C in a water bath for one hour while being constantly stirred. A double-layered Whatman Number 1 filter paper was used to quantitatively filter the extract, and 50% methanol was used to rinse it. Twenty milliliters of distilled water, 2.5 milliliters of Folin-Denis reagent, and
10 milliliters of 17% Na2CO3 were added to one milliliter of sample extract to produce a bluish-green color. The mixture was then let to stand for 20 minutes. The amount of tannin was determined by comparing the absorbance, which was measured at 760 nm, with a standard curve that was constructed in the range of 0-10 ppm.
Determination of saponins: The Saponin analysis was carried out using the Mujeeb et al.16 method. One gram of the finely powdered sample was mixed with precisely 100 milliliters of isobutyl alcohol and swirled for 5 hours. After adding 20 milliliters of a 40% saturated magnesium carbonate solution, the liquid was filtered. As 1 mL of colorless solution was mixed with 2 mL of 5% FeCl3 solution and 50 mL of distilled water, and the mixture was left for 30 minutes to develop color (blood red). At 380 nm, the absorbance of the samples and the standard was measured and expressed in milligrams per gram. A reference range of 0-10 ppm was used to prepare the standard saponin solution.
Determination of antioxidant activity using the ferric reducing/antioxidant power (FRAP): The FRAP assay was conducted following the method described by Chaves et al.17. Aliquots of 0.2 mL of sample (at four different concentrations: 0.1, 0.5, 1, and 2 mg/mL; two replicates per sample and concentration) had 3.8 mL of FRAP reagent added. This reagent was previously prepared by mixing 10 parts of 300 mM sodium acetate buffer solution at pH 3.6, 1 part of 10 mM TPZT, and 1 part of 20 mM FeCl3hexahydrate. The resulting mix was incubated for 30 min at 37°C. The absorbance increase was measured at 593 nm in a UV-30 spectrophotometer. The blank was prepared by substituting the same amount of diluted extract with methanol. The results were expressed in milligram equivalents of FeSO4per milligram of dry weight.
Hydrogen peroxide scavenging activity: The ability ofthe herbal mixtureto scavenge hydrogen peroxide was determined according to the method of Güder and Korkmaz18. Hydrogen peroxide solution (40 mM) was prepared in phosphate buffer (pH 7.4). Herbal mixture extract (100 μg/mL) in distilled water was added to hydrogen peroxide solution (0.6 mL, 40 mM). Absorbance of hydrogen peroxide at 230 nm was determined against a blank solution containing the phosphate buffer without hydrogen peroxide.
| Ac | = | Absorbance of control that contains H2O2 and PB | |
| As | = | Absorbance of sample |
Determination of antioxidant activity by DPPH: The antioxidant activity of fractions of methanol extract of the plant was assayed by the 1,1-Diphenyl-2-picrylhydrazyl (DPPH) radical scavenging method described by Karadag et al.19.
Principle: 1,1-Diphenyl-2-picrylhydrazyl (DPPH) contains an odd electron in its structure. Its deep purple colour is reduced to a colourless compound, 2,2-diphenyl-1-picryhydrazine, when it reacts with an antioxidant, which can donate a hydrogen atom or an electron to it. The colour change was measured spectrophotometrically at 517 nm using a UV/Visible light spectrophotometer.
The assay mixture contained 2 mL of 1.0 mM DPPH radical solution prepared in methanol and 1 mL of standard or extract solution of different concentrations (10-500 μg/mL). The solution was rapidly mixed and incubated in the dark at 37°C for 20 min. The decrease in absorbance of each solution was measured at 517 nm using a spectrophotometer. Ascorbic acid was used as a positive control, while 2 mL of 1.0 mM DPPH radical solution with 1 mL of ethanol was taken as a blank.
The percentage of radical scavenging (%) was calculated by:
Where:
| Ac | = | Absorbance of control at 517 nm | |
| As | = | Absorbance of sample at 517 nm |
The concentration of sample required to scavenge 50% of DPPH free radical (IC50) was determined from the curve of percentage inhibition plotted against the respective concentrations.
β-carotene bleaching inhibition assay: In this assay, antioxidant activity was determined by measuring the inhibition of conjugated dienehydroperoxides arising from linoleic acid oxidation20.
A stock solution of β-Carotene/Linoleic acid mixture was prepared as follows: About 4 mg of β-Carotene was weighed and dissolved in 2 mL of chloroform. Exactly 50 μL of linoleic acid was added, followed by 400 μL of Tween-40. The solution was mixed thoroughly and kept in a hot air oven to evaporate chloroform from the mixture. About 200 mL of distilled water was added. The test for β-Carotene was taken as follows.
Exactly 5 mL of stock solution was added to 100 μL of each fraction. It was incubated for 120 minutes at 50°C. The absorbance was measured at 520 nm. It was zeroed using distilled water, while 5 mL of the stock solution was taken as absorbance of the blank.
Where:
| AS120 | = | Absorbance of test sample after incubation for 120 minutes | |
| AC120 | = | Absorbance of control after incubation for 120 minutes | |
| AC0 | = | Absorbance of control before incubation for 120 minutes |
Statistical analysis: The data were analysed by the Analysis of Variance (ANOVA) using SPSS program (version 20 SPSS Inc., Chicago, IL, USA). The differences in parameters between the various animal groups were compared using the Duncan multiple comparison test (post hoc test). The results were expressed as Mean±Standard Deviation (SD). A p value less than 0.05 was considered as significant (p<0.05). Results were presented in charts using Microsoft Excel.
RESULTS
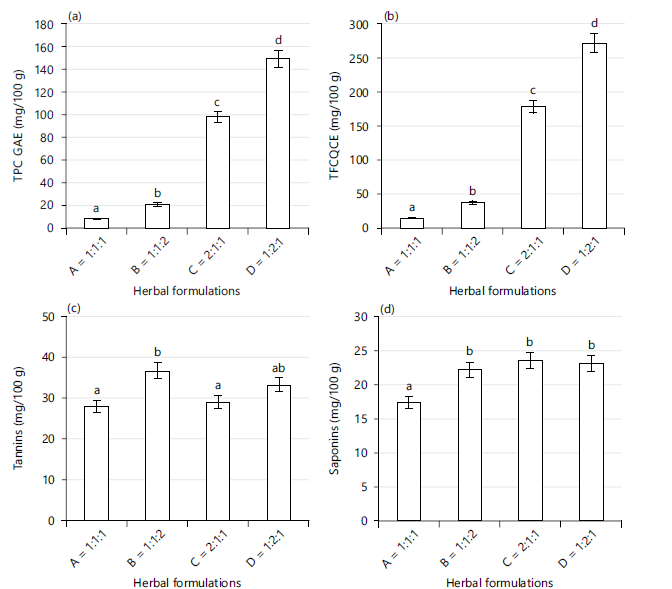
Phytochemical constituents of herbal formulation of Carica papaya, Azadirachta indica and Allium sativa (A-1:1:1, B-1:1:2, C-2:1:1, and D-1:2:1): The result of phytochemical constituents of different ratios of Carica papaya: Azadirachta indica: Allium sativa herbal formulation is represented in Fig. 1(a-d). Total phenolic content shows a decreasing trend as D>C>B>A (ie 149.5>98.25>20.78>8.47 GAE mg/100 g) across the different ratios in Fig. 1a with D displaying the highest value of total phenolic contengt. Also, a similar trend was noticed with the flavonoid content, D>C>B>A (ie 271.95>178.77>37.91>15.54 QCE mg/100 g) across the different ratios as seen in Fig. 1b. The presence of tannins (Fig. 1c) and saponins (Fig. 1d) did not vary greatly across the different ratios of the formulations. However, B tends to be higher (36.58 mg/mL) in tannins as shown in Fig. 1c while C was higher (23.63 mg/mL) in saponins as displayed in Fig. 1d. The result puts it obviously that flavonoids and phenolics were more abundant in the herbal formulation of Carica papaya: Azadirachta indica: Allium sativa.
|
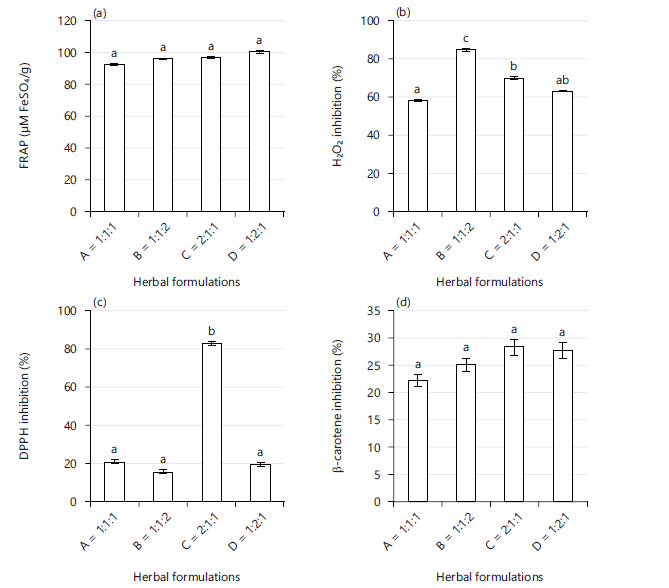
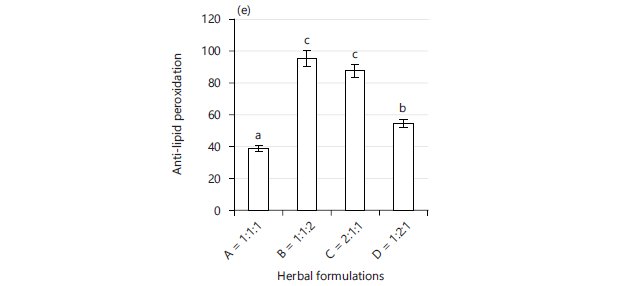
Antioxidant potential of herbal formulation of Carica papaya, Azadirachta indica, and Allium sativa (A-1:1:1, B-1:1:2, C-2:1:1, and D-1:2:1): Figure 2(a-e) presents the result of different antioxidant potentials of different ratios of Carica papaya: Azadirachta indica: Allium sativa herbal formulation. All the ratios showed reasonable ferric reducing antioxidant power (FRAP), hydrogen peroxide, and anti-lipid peroxidation inhibition capacities. There was a decreasing trend that followed the order of D>C>B>A in the FRAP concentration as presented in Fig. 2a. The percentage inhibitions of hydrogen peroxide in Fig. 2b show that A, B, and C were closely related, while B displayed the highest value of 84.83%. In Fig. 2c, the DPPH scavenging activity was clearly higher in C (83.1%) as compared to other ratios. The β-carotene bleaching inhibition did not show significant variation among the ratios, as shown in Fig. 2d. Anti-lipid peroxidation activity of B, C and D was above 50% except for A, which was 38.58% as shown in Fig. 2e. There was a moderate performance in 2,2-Diphenyl-1-picrylhydrazyl (DPPH) and β-carotene bleaching inhibition antioxidant activity except for ratio C-2:1:1 that displayed excellent DPPH activity as compared to the other ratios of the herbal formulation in the present study.
|
DISCUSSION
Phytochemicals are bioactive compounds of plant origin that are usually secondary metabolites, which are crucial for therapeutic effects like antioxidantandanti-canceractivities.Analyzing these constituents provides a scientific basis for traditional medicinal uses, reveals synergistic interactions, and identifies potential adulteration21,22. Studies often involve qualitative and quantitative screening to determine the presence and concentrations of these compounds to substantiate evidence for the efficacy and safety of herbal products. The present result obtained through spectrophotometric assay shows that the various herbal combinations of Carica papaya, Azadirachta indica, and Allium sativa are rich in phenolics, flavonoids, tannins, and saponins.
According to earlier studies, flavonoids and phenolics have a variety of biological actions, such as cytotoxic, anti-inflammatory, antiviral, antibacterial, antiallergic, and anticancer effects23,24. Saponins have a wide range of biological activities, such as lowering serum cholesterol, increasing cell membrane permeability, abortifacient properties, immunomodulatory properties, cytotoxic effects on malignant tumor cells, and participating in the synergistic enhancement of immunotoxin toxicity25 and anti-diabetic properties26. Tanning agents have been used to treat non-insulin-dependent diabetes mellitus by increasing glucose absorption and decreasing abiogenesis. Additionally, they have been shown to possess anti-inflammatory, anti-carcinogenic, anti-mutagenic, and cardioprotective qualities.
Thus, the result of the present investigation proved that the presence of various phytochemicals such as phenolic compounds, flavonoids, saponins, and tannins in the herbal formulations of Carica papaya, Azadirachta indica, and Allium sativa may be responsible for pharmacological actions24. Therefore, it can be affirmed that the polyphenolic content of the herbal mixtures would indicate their effective antioxidant potentials because the phenolic constituents can react with active oxygen radicals such as hydroxyl radical, superoxide anion radical and lipid peroxy radical27. Concomitantly, previous reports showed that there is high correlation between antioxidant activity and polyphenolics content28. Similarly, the current finding is in tandem with reports of Sharma et al.5, Rahmani et al.7 and Tudu et al.10 where the individual plant that formed the component of the herbal formulations were reported to contain phytochemicals revealed by the current investigation. However, report was not found on the phytoconstituents of herbal mixtures of Carica papaya, Azadirachta indica, and Allium sativa.
Since it is generally acknowledged that lipid peroxidation occurs via the free radical chain reaction, radical scavengers can directly interact with and quench peroxide radicals to stop peroxidation chain events, which are important in the etiology of many diseases29. The reducing potential of an antioxidant reacting with a ferric tripyridyltriazine (Fe3+-TPTZ) complex and generating a colored ferrous tripyridyltriazine (Fe2+-TPTZ) is measured by the ferric reducing antioxidant power (FRAP) assay.
The free radical chain breaking takes place through donating a hydrogen atom30. Thus, the evident values of FRAP recorded in the herbal formulations show that the antioxidants present in the samples have H-donating ability and may also contain methoxy and keto groups, triterpenes and acid groups31 relevant to the FRAP antioxidant activity observed in the present study. The hydrogen peroxide (H2O2) inhibition antioxidant assaymeasures a substance’s ability to neutralize H2O2, a reactive oxygen species (ROS) involved in oxidative damage, by quantifying the decrease in H2O2 concentration over time32. Although hydrogen peroxide may not be very reactive, however, it can be toxic because it may produce hydroxyl radicals in the cells33, thus, removing H2O2 is very important in the cell. The scavenging ability of the herbal formulations in the present evaluation showed a powerful activity on hydrogen peroxide and decreases its concentration which is an indication of H2O2 removal potential34.
Since the radical molecules can react directly with antioxidants27, the assay based on the use of DPPH radicals is one of the most widely used spectrophotometric methods for determining the antioxidant capacity of plant extracts, meals, drinks, and vegetable extracts. Additionally, because the DPPH scavenging method is straightforward, quick, sensitive, and repeatable, it has been utilized to assess the antioxidant activity of compounds35. According to the current findings regarding the herbal formulations’ capacity to inhibit DPP, they appear to be effective free radical scavengers, particularly those of the peroxy type, and they may also be able to prevent lipid autoxidation, which could help treat a number of illnesses where lipid peroxidation is a key pathophysiological mechanism.This further claims that the herbal formulations contain substances that can donate hydrogen to a free radical in order to eliminate the odd electron that causes the radical’s reactivity. This suggests that the herbal formulations could help treat pathological damage caused by radicals, particularly at varying concentrations36.
In contrast, the β-carotene bleaching method relies on the fact that β-carotene loses its yellow color as a result of reacting with radicals created by the oxidation of linoleic acid in an emulsion37. Antioxidants can slow down the rate at which β-carotene bleaching occurs.
The results of the present study implicated the herbal formulations to have displayed β-carotene bleaching capacity and may have occurred as a result of antioxidative phytochemicals present in the herbal formulations. Conversely, the anti-lipid peroxidation inhibition assayevaluates how well the herbal extract protects lipids from oxidative damage by measuring the production of malondialdehyde (MDA) or other lipid peroxidation products38
The ability of herbal preparations to scavenge hydroxyl radicals and hence reduce lipid peroxidation is closely linked to their antioxidant potential39. Lipid hydroperoxides have the ability to bond with DNA, causing strand breakage, carcinogenesis, and mutation40, and they can harm any molecule in the biological system. Because hemoglobin and polyunsaturated fatty acids are heavily accumulated, erythrocytes are inherently more susceptible to peroxidation. Erythrocytes are constantly exposed to high oxygen tension during breathing, which can cause oxidative damage41. Additionally, when erythrocytes are exposed to toxicants, free radicals are produced, which may cause membrane damage and hemolysis42. The herbal formulations used in this investigation demonstrated scavenging action against hydroxyl radicals, indicating the existence of primary antioxidants with the ability to prevent lipid peroxidation and hemolysis7.
Furthermore, the results of in vitro antioxidant analysis recorded in the present investigation about the herbal formulation of Carica papaya, Azadirachta indica and Allium sativa are in concomitance with results of individual component of the herbal formulations that were presented in the reports of Sharma et al.5, Rahmani et al.7 and Tudu et al.10. Although, no report on antioxidant potential of the herbal combinations investigated in the present study was found.
CONCLUSION
Based on the observations from the present study, it can be concluded that the various ratios of ethanol extract of herbal formulation containing Carica papaya, Azadirachta indica, and Allium sativa exhibited pronounced antioxidant activity supported by the presence of potent free radical scavenging and biologically active compounds. However, the herbal mixture containing Carica papaya, Azadirachta indica, and Allium sativa in the ratio of 2:1:1 w/w, respectively was consistently viable in terms of photoconstituets and antioxidant activity as determined when compared to the 1:1:1, 1:2:1 and 1:1:2 w/w ratios and hence was selected for fractionation.
SIGNIFICANCE STATEMENT
This study shows that herbal formulations of Carica papaya, Azadirachta indica, and Allium sativa affect the compositions and antioxidant activity of the mixture. The present investigation reveals that the herbal formulation of Carica papaya, Azadirachta indica, and Allium in the ratio of 2:1:1 w/w, respectively, was consistently viable in terms of photochemical and antioxidant activity. By implication, this study has opened up an opportunity for the utilization of a herbal combination of Carica papaya, Azadirachta indica, and Allium sativa in the treatment and management of oxidative stress-related pathological conditioms.
REFERENCES
- Davis, C.C. and P. Choisy, 2024. Medicinal plants meet modern biodiversity science. Curr. Biol., 34: R158-R173.
- Lonkala, S. and A.R.N. Reddy, 2019. Antibacterial activity of Carica papaya leaves and Allium sativum cloves alone and in combination against multiple strains. Pharmacogn. J., 11: 600-602.
- Dharmarathna, S.L.C.A., S. Wickramasinghe, R.N. Waduge, R.P.V.J. Rajapakse and S.A.M. Kularatne, 2013. Does Carica papaya leaf-extract increase the platelet count? An experimental study in a murine model. Asian Pac. J. Trop. Biomed., 3: 720-724.
- Otsuki, N., N.H. Dang, E. Kumagai, A. Kondo, S. Iwata and C. Morimoto, 2010. Aqueous extract of Carica papaya leaves exhibits anti-tumor activity and immunomodulatory effects. J. Ethnopharmacol., 127: 760-767.
- Sharma, A., R. Sharma, M. Sharma, M. Kumar and M.D. Barbhai et al., 2022. Carica papaya L. leaves: Deciphering its antioxidant bioactives, biological activities, innovative products, and safety aspects. Oxid. Med. Cell. Longevity, 2022.
- Panzarini, E., M. Dwikat, S. Mariano, C. Vergallo and L. Dini, 2014. Administration dependent antioxidant effect of Carica papaya seeds water extract. Evidence-Based Complementary Altern. Med., 2014.
- Rahmani, A.H., A. Almatroudi, F. Alrumaihi and A.A. Khan, 2018. Pharmacological and therapeutic potential of neem (Azadirachta indica). Pharmacogn. Rev., 12: 250-255.
- Islas, J.F., E. Acosta, Z. G-Buentello, J.L. Delgado-Gallegos, M.G. Moreno-Treviño, B. Escalante and J.E. Moreno-Cuevas, 2020. An overview of neem (Azadirachta indica) and its potential impact on health. J. Funct. Foods, 74.
- Omóbòwálé, T.O., A.A. Oyagbemi, O.A. Adejumobi, E.V. Orherhe and A.S. Amid et al., 2016. Preconditioning with Azadirachta indica ameliorates cardiorenal dysfunction through reduction in oxidative stress and extracellular signal regulated protein kinase signalling. J. Ayurveda Integr. Med., 7: 209-217.
- Tudu, C.K., T. Dutta, M. Ghorai, P. Biswas and D. Samanta et al., 2022. Traditional uses, phytochemistry, pharmacology and toxicology of garlic (Allium sativum), a storehouse of diverse phytochemicals: A review of research from the last decade focusing on health and nutritional implications. Front. Nutr., 9.
- Zeng, Y., Y. Li, J. Yang, X. Pu and J. Du et al., 2017. Therapeutic role of functional components in Alliums for preventive chronic disease in human being. Evidence-Based Complementary Altern. Med., 2017.
- Batiha, G.E.S., A.M. Beshbishy, L.G. Wasef, Y.H.A. Elewa and A.A. Al-Sagan et al., 2020. Chemical constituents and pharmacological activities of garlic (Allium sativum L.): A review. Nutrients, 12.
- Molole, G.J., A. Gure and N. Abdissa, 2022. Determination of total phenolic content and antioxidant activity of Commiphora mollis (Oliv.) Engl. resin. BMC Chem., 16.
- Kalita, P., B.K. Tapan, T.K. Pal and R. Kalita, 2013. Estimation of total flavonoids content (TFC) and anti oxidant activities of methanolic whole plant extract of Biophytum sensitivum Linn. J. Drug Delivery Ther., 3: 33-37.
- Sharma, T., R. Gamit, R. Acharya and V.J. Shukla, 2021. Quantitative estimation of total tannin, alkaloid, phenolic, and flavonoid content of the root, leaf, and whole plant of Byttneria herbacea Roxb. AYU, 42: 143-147.
- Mujeeb, F., P. Bajpai and N. Pathak, 2014. Phytochemical evaluation, antimicrobial activity, and determination of bioactive components from leaves of Aegle marmelos. BioMed Res. Int., 2014.
- Chaves, N., M. Navarro, M. Flores and J.C. Alias, 2025. Correlation between the amount of total phenolic contents and antioxidant activity of mediterranean shrubs: Are phenols a good indicator of the antioxidant activity of these species? Nat. Prod. J., 15.
- Güder, A. and H. Korkmaz, 2012. Evaluation of in-vitro antioxidant properties of hydroalcoholic solution extracts Urtica dioica L., Malva neglecta Wallr. and their mixture. Iran. J. Pharm. Res., 11: 913-923.
- Karadag, A., B. Ozcelik and S. Saner, 2009. Review of methods to determine antioxidant capacities. Food Anal. Methods, 2: 41-60.
- Ahmed, D., W. Malik, M. Maqsood, I. Atique and M.T. Qamar, 2022. Study of anti-diabetic, beta-carotene-bleaching inhibiting and iron chelating properties of Carissa opaca root extracts. Braz. J. Pharm. Sci., 58.
- Arya, V., S. Yadav, S. Kumar and J.P. Yadav, 2011. Antioxidant activity of organic and aqueous leaf extracts of Cassia occidentalis L. in relation to their phenolic content. Nat. Prod. Res., 25: 1473-1479.
- Zafar, F., H.M. Asif, G. Shaheen, A.O. Ghauri and S.R. Rajpoot et al., 2023. A comprehensive review on medicinal plants possessing antioxidant potential. Clin. Exp. Pharmacol. Physiol., 50: 205-217.
- Yakub, O.E., M.R.C. Schetinge, K.A. Arowora and C.O. Shaibu, 2022. GC-MS characterization and antioxidant properties of partially purified ethanol extract of Nauclea latifolia (African peach) stem bark. Biotechnology, 21: 146-155.
- Saba, A., N. Rehan and S.S. Aziz, 2023. Phytochemical analysis of unani herbal formulation “Mazher-Ul-Ajaib”. J. Pharmacogn. Phytochem., 12: 105-117.
- Thakur, M., M.F. Melzig, H. Fuchs and A. Weng, 2011. Chemistry and pharmacology of saponins: Special focus on cytotoxic properties. Botanics: Targets Ther., 1: 19-29.
- Zheng, T., G. Shu, Z. Yang, S. Mo, Y. Zhao and Z. Mei, 2012. Antidiabetic effect of total saponins from Entada phaseoloides (L.) Merr. in type 2 diabetic rats. J. Ethnopharmacol., 139: 814-821.
- Aliyu, A.B., M.A. Ibrahim, A.M. Musa, A.O. Musa, J.J. Kiplimo and A.O. Oyewale, 2013. Free radical scavenging and total antioxidant capacity of root extracts of Anchomanes difformis Engl. (Araceae). Acta. Pol. Pharm., 70: 115-121.
- El Rayess, Y., L. Nehme, C. Ghanem, M. El Beyrouthy, C. Sadaka et al., 2023. Phenolic content, antioxidant and antimicrobial activities evaluation and relationship of commercial spices in the lebanese market. BMC Chem., 17.
- Valgimigli, L., 2023. Lipid peroxidation and antioxidant protection. Biomolecules, 13.
- Rajurkar, N.S. and S.M. Hande, 2011. Estimation of phytochemical content and antioxidant activity of some selected traditional Indian medicinal plants. Indian J. Pharm. Sci., 73: 146-151.
- Shukla, A., S. Vats and R.K. Shukla, 2015. Phytochemical screening, proximate analysis and antioxidant activity of Dracaena reflexa lam. leaves. Indian J. Pharm. Sci., 77: 640-644.
- Jomova, K., R. Raptova, S.Y. Alomar, S.H. Alwasel, E. Nepovimova, K. Kuca and M. Valko, 2023. Reactive oxygen species, toxicity, oxidative stress, and antioxidants: Chronic diseases and aging. Arch. Toxicol., 97: 2499-2574.
- Lenzen, S., V.I. Lushchak and F. Scholz, 2022. The pro-radical hydrogen peroxide as a stable hydroxyl radical distributor: Lessons from pancreatic beta cells. Arch. Toxicol., 96: 1915-1920.
- Mohammedi, Z., 2021. Extraction, phenolic content and hydrogen peroxide scavenging capacity of extracts from some honey samples, propolis and bee pollen. Biol. Med. Nat. Prod. Chem., 10: 27-32.
- Gulcin, İ. and S.H. Alwasel, 2023. DPPH radical scavenging assay. Processes, 11.
- Aiyegoro, O.A. and A.I. Okoh, 2010. Preliminary phytochemical screening and in vitro antioxidant activities of the aqueous extract of Helichrysum longifolium DC. BMC Complementary Altern. Med.
- Lu, Y., T.J. Knoo and C. Wiart, 2014. Phytochemical analysis and antioxidant activity determination on crude extracts of Melodinus eugeniifolus barks and leaves from Malaysia. Pharmacol. Pharm., 5: 773-780.
- Félix, R., P. Valentão, P.B. Andrade, C. Félix, S.C. Novais and M.F.L. Lemos, 2020. Evaluating the in vitro potential of natural extracts to protect lipids from oxidative damage. Antioxidants, 9.
- Yap, V.L., L.F. Tan, M. Rajagopal, C. Wiart, M. Selvaraja, M.Y. Leong and P.L. Tan, 2023. Evaluation of phytochemicals and antioxidant potential of a new polyherbal formulation TC-16: Additive, synergistic or antagonistic? BMC Complementary Med. Ther., 23.
- Chandimali, N., S.G. Bak, E.H. Park, H.J. Lim and Y.S. Won et al., 2025. Free radicals and their impact on health and antioxidant defenses: A review. Cell Death Discovery, 11.
- Shabbir, M., M.R. Khan and N. Saeed, 2013. Assessment of phytochemicals, antioxidant, anti-lipid peroxidation and anti-hemolytic activity of extract and various fractions of Maytenus royleanus leaves. BMC Complementary Altern. Med., l. 13.
- Orrico, F., S. Laurance, A.C. Lopez, S.D. Lefevre, L. Thomson, M.N. Möller and M.A. Ostuni, 2023. Oxidative stress in healthy and pathological red blood cells. Biomolecules, 13.
How to Cite this paper?
APA-7 Style
Abu,
M.S., Yakubu,
O.E., Tatah,
S.V., Danka,
Y.J. (2026). Phytochemical Profiling and Antioxidant Assessment of a Herbal Formulation Containing Carica papaya, Azadirachta indica, and Allium sativum. Science International, 14(1), 29-40. https://doi.org/10.17311/sciintl.2026.29.40
ACS Style
Abu,
M.S.; Yakubu,
O.E.; Tatah,
S.V.; Danka,
Y.J. Phytochemical Profiling and Antioxidant Assessment of a Herbal Formulation Containing Carica papaya, Azadirachta indica, and Allium sativum. Sci. Int 2026, 14, 29-40. https://doi.org/10.17311/sciintl.2026.29.40
AMA Style
Abu
MS, Yakubu
OE, Tatah
SV, Danka
YJ. Phytochemical Profiling and Antioxidant Assessment of a Herbal Formulation Containing Carica papaya, Azadirachta indica, and Allium sativum. Science International. 2026; 14(1): 29-40. https://doi.org/10.17311/sciintl.2026.29.40
Chicago/Turabian Style
Abu, Michael, Sunday, Ojochenemi Ejeh Yakubu, Silas Verwiyeh Tatah, and Yab Jeremiah Danka.
2026. "Phytochemical Profiling and Antioxidant Assessment of a Herbal Formulation Containing Carica papaya, Azadirachta indica, and Allium sativum" Science International 14, no. 1: 29-40. https://doi.org/10.17311/sciintl.2026.29.40

This work is licensed under a Creative Commons Attribution 4.0 International License.


